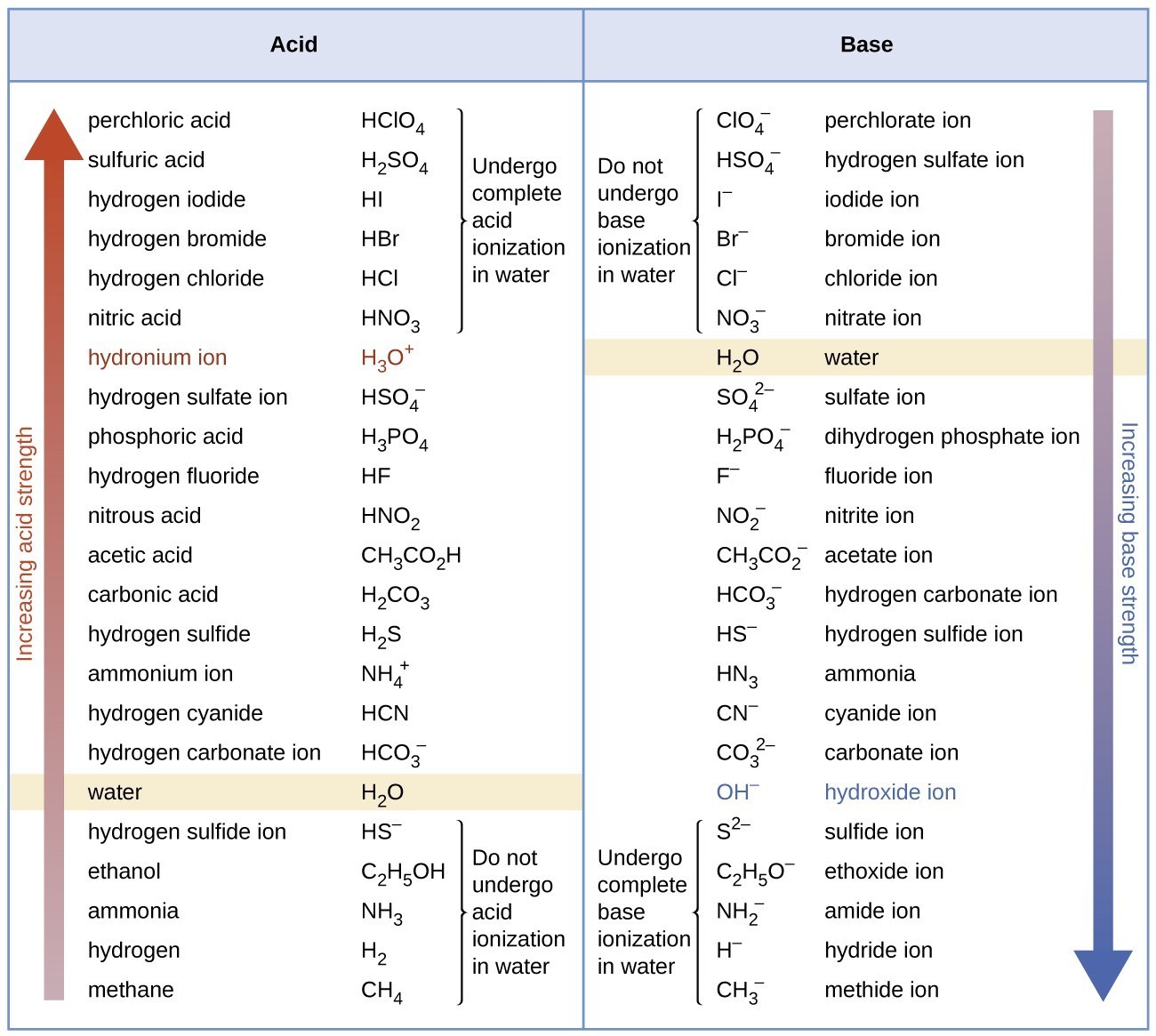
Table 2 from Dissociation Constant of N-(2-Acetamido)-Iminodiacetic Acid Monosodium (ADA) from (278.15 to 328.15) K | Semantic Scholar

Table 1 from Ionization constant of 5-5'-diethylbarbituric acid from 0-degrees to 60-degrees-C | Semantic Scholar

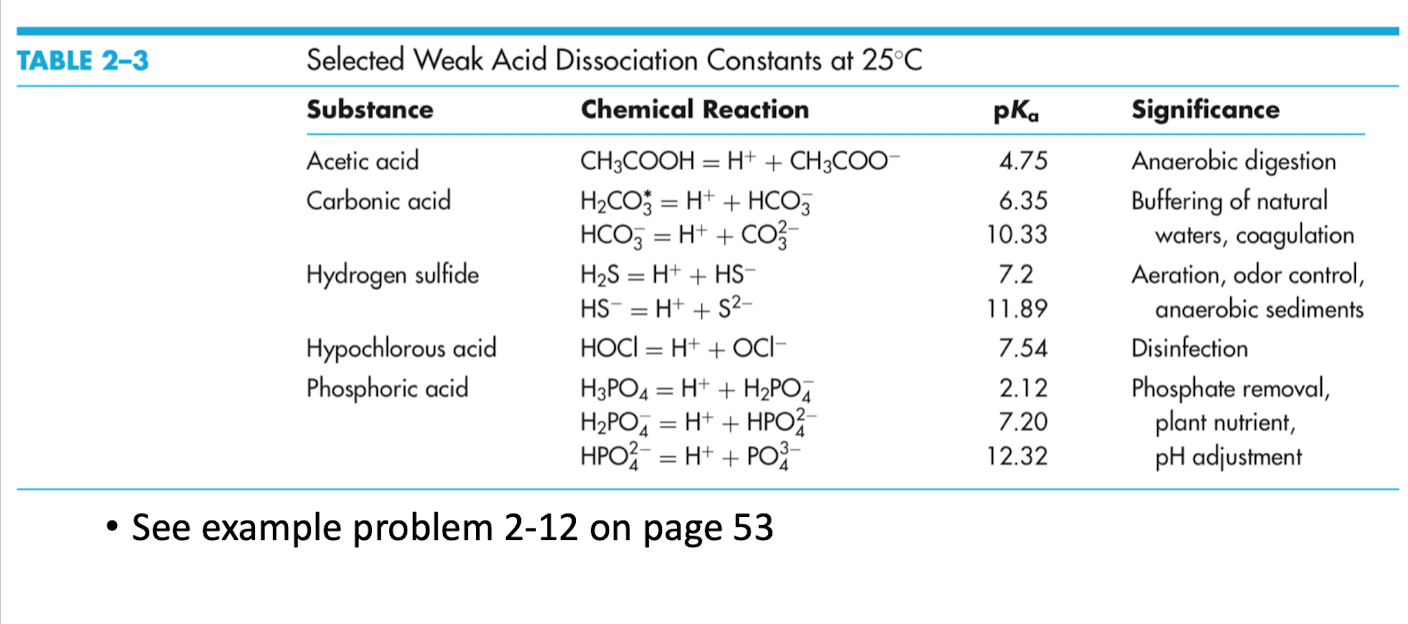
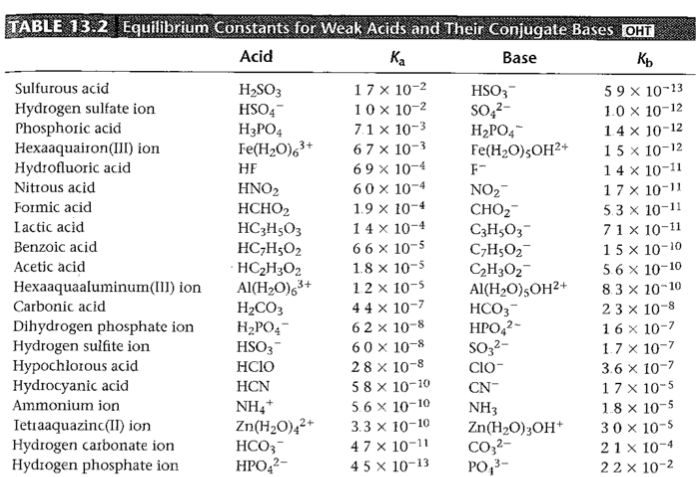
Table 10.4 from 10 Acid-base Interactions 10 . 1 GENERAL CONCEPT OF ACID-BASE INTERACTIONS George Wypych | Semantic Scholar
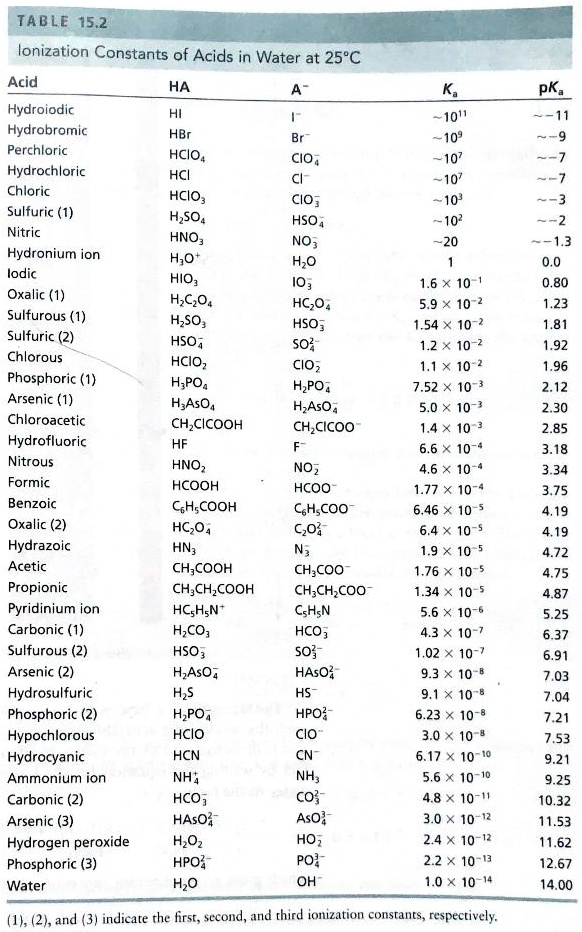
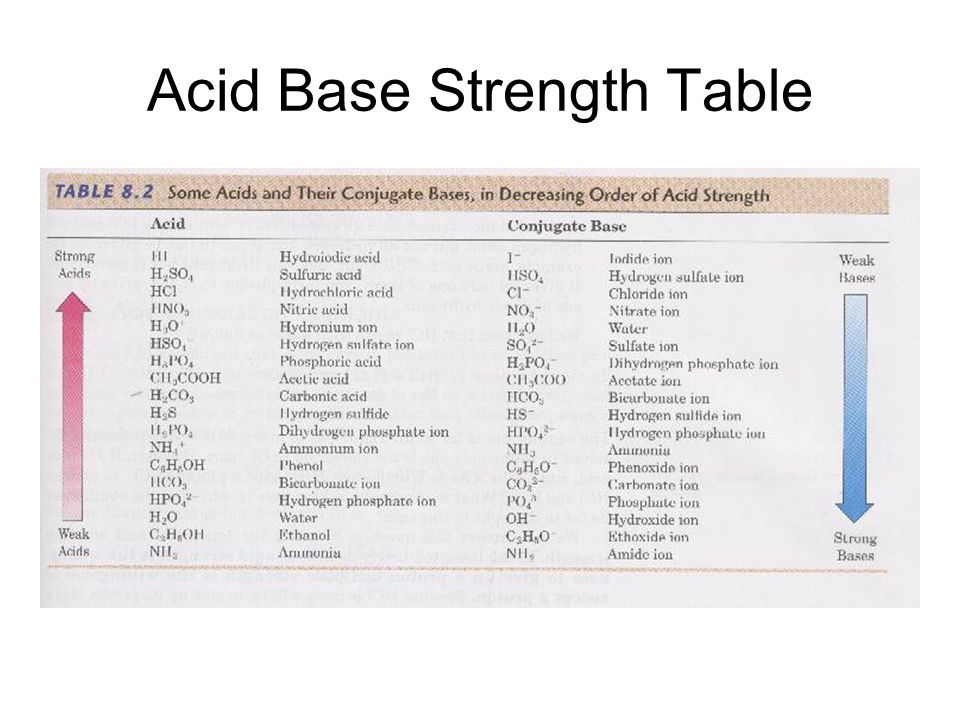
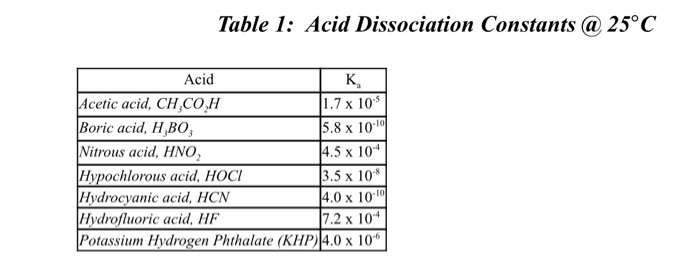
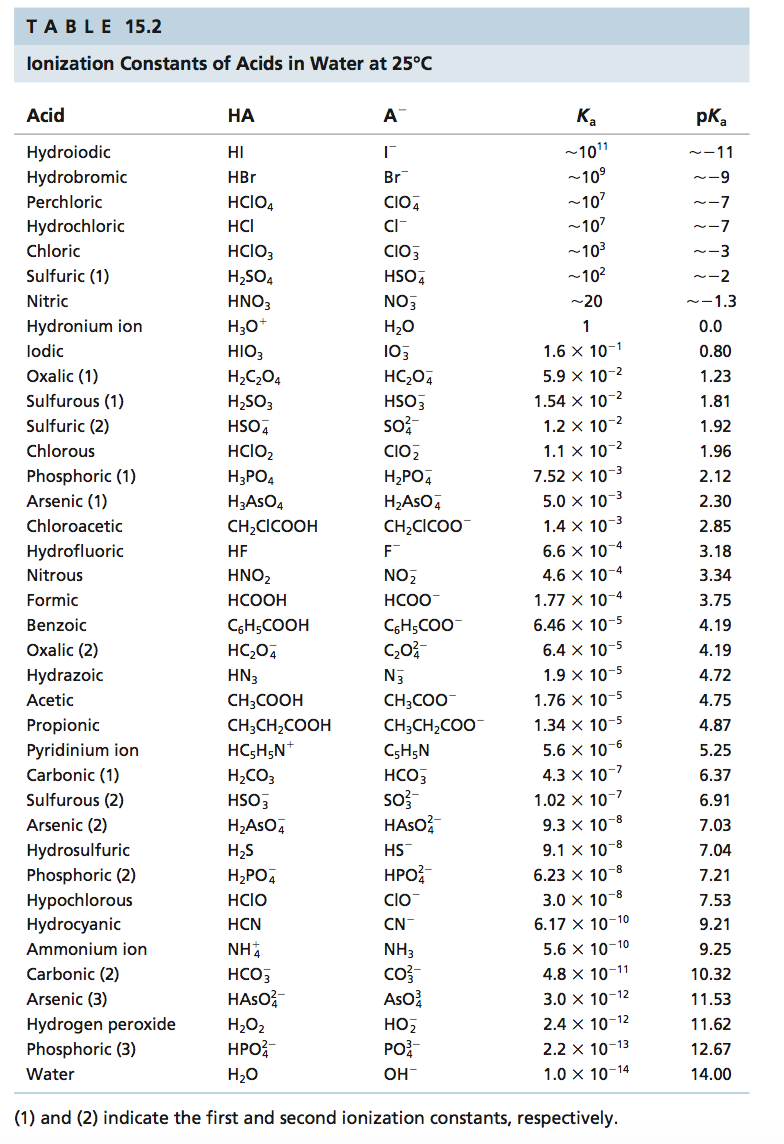
SOLVED: TABLE 15.2 Ionization Constants of Acids in Water at 25"C Acid HA Hydroiodic Hydrobromic HBr Perchloric HCIO4 CIO, Hydrochloric HCI Chloric HCIO; CIO; Sulfuric (1) HSO4 HSO, Nitric HNO; NO; Hydronium
![PDF] Determination of Dissociation Constants of Malonic Acid in (Ethylene Glycol-Water)X% Mixed Solvent at Different Temperatures Using Electromotive Force Measurements | Semantic Scholar PDF] Determination of Dissociation Constants of Malonic Acid in (Ethylene Glycol-Water)X% Mixed Solvent at Different Temperatures Using Electromotive Force Measurements | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6d04a3862b2f2dbb01b9220329429f516e4430e9/2-Table1-1.png)
PDF] Determination of Dissociation Constants of Malonic Acid in (Ethylene Glycol-Water)X% Mixed Solvent at Different Temperatures Using Electromotive Force Measurements | Semantic Scholar

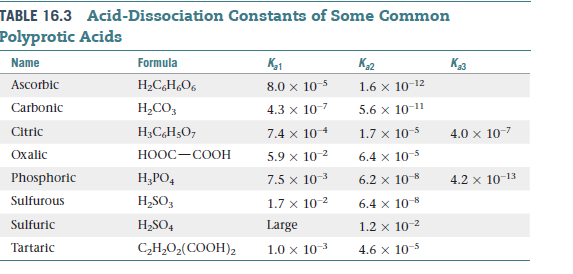
Table 3 from Measurement of dissociation constants (pKa values) of organic compounds by multiplexed capillary electrophoresis using aqueous and cosolvent buffers. | Semantic Scholar
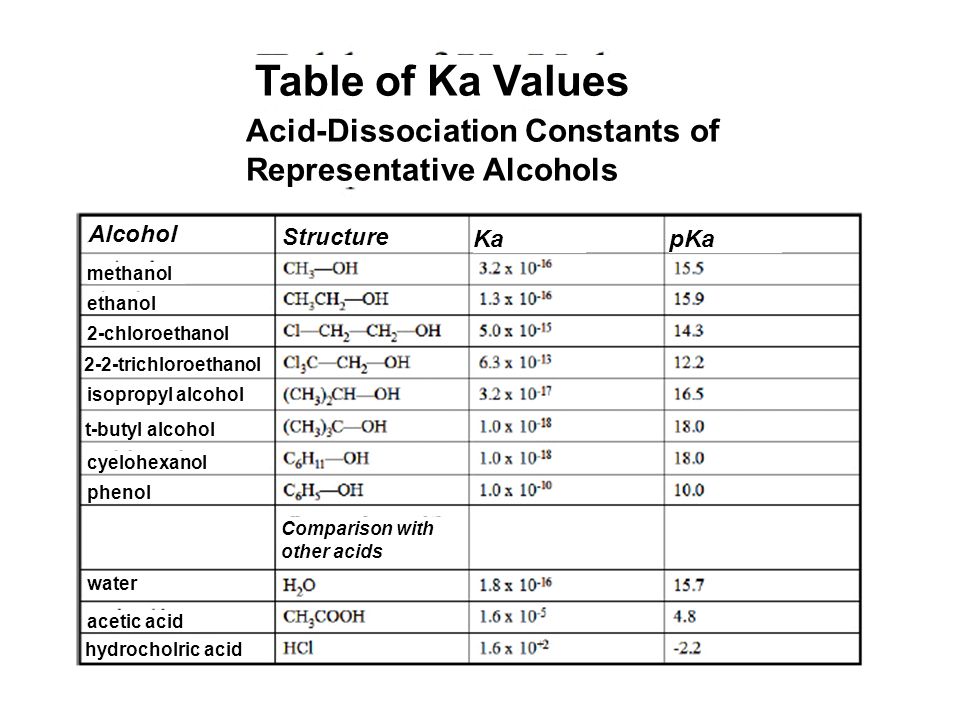
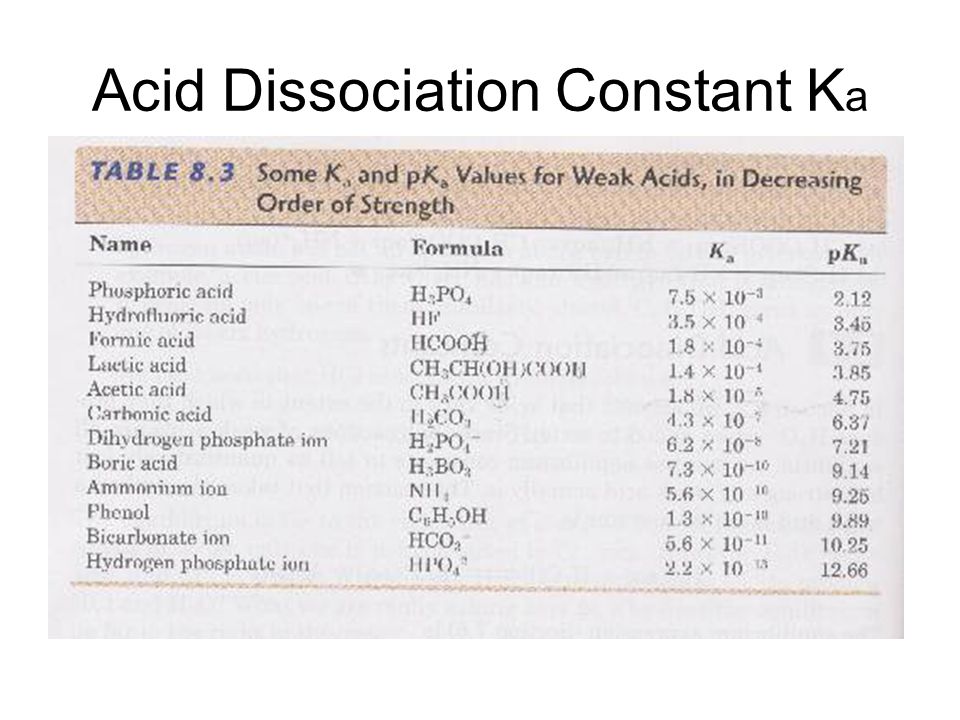
Table of Ka Values Acid-Dissociation Constants of Representative Alcohols Alcohol Structure Ka pKa methanol ethanol 2-chloroethanol 2-2-trichloroethanol. - ppt video online download

Table 2 from Prediction of electrophoretic mobilities. Part 2.† Effect of acid dissociation constant on the intrinsic mobilities of aliphatic carboxylates and amines | Semantic Scholar